The Department of Health (DH) today (November 12) conducted an operation against the sale of unregistered pharmaceutical products labelled in Japanese, during which a 31-year-old woman was arrested for suspected illegal sale of unregistered pharmaceutical products and Part 1 poisons.
Acting upon a public complaint, unregistered pharmaceutical products labelled in Japanese were found being offered for sale via a social media platform. Two products that did not bear Hong Kong pharmaceutical product registration numbers were seized during today’s operation.
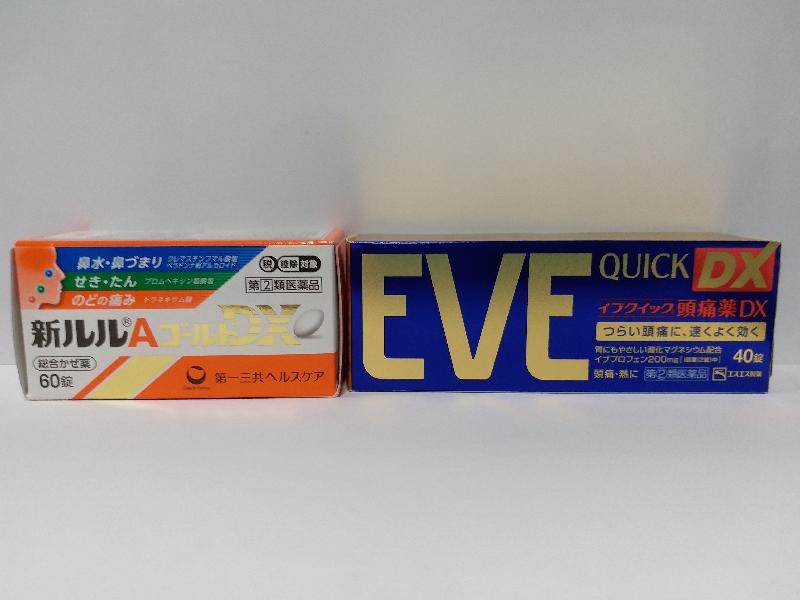
One of the products is a cold and flu medication labelled in Japanese. It is believed to contain tranexamic acid, dihydrocodeine and methylephedrine, which are all Part 1 poisons under the Pharmacy and Poisons Ordinance (PPO)(Cap 138). Tranexamic acid is used in the treatment of haemorrhage and can cause gastrointestinal disturbances, and inappropriate use may cause cerebral thrombosis. Dihydrocodeine is an opioid analgesic used as a cough suppressant and for pain relief, and its side effects include nausea, vomiting, constipation and drowsiness. Methylephedrine is used for nasal congestion and cough, and it may cause tachycardia, anxiety, restlessness and insomnia.
The other product is named Eve Quick DX and is believed to contain the Part 1 poison ibuprofen. Ibuprofen is a non-steroidal anti-inflammatory drug for pain relief with side effects including gastrointestinal bleeding.
The DH’s investigation is ongoing.
According to the PPO, all pharmaceutical products must be registered with the Pharmacy and Poisons Board of Hong Kong before they can be legally sold in the market. Part I poisons should be sold at pharmacies under the supervision of registered pharmacists. Illegal sale or possession of unregistered pharmaceutical products and Part 1 poisons are criminal offences. The maximum penalty for each offence is a fine of $100,000 and two years’ imprisonment.
Members of the public should not buy or consume unregistered pharmaceutical products as they may not comply with the safety, efficacy and quality requirements. All registered pharmaceutical products should carry a Hong Kong registration number on the package in the format of “HK-XXXXX”. People should seek advice from healthcare professionals before taking medications. They should not purchase medicines overseas and resell them in Hong Kong.
People who have purchased the above products should stop using them and consult healthcare professionals for advice if they are feeling unwell. They can submit the products to the DH’s Drug Office at Room 1856, Wu Chung House, 213 Queen’s Road East, Wan Chai, during office hours for disposal.